Articles
- Page Path
- HOME > Kosin Med J > Volume 34(1); 2019 > Article
-
Case Report
Mycoplasma Pneumoniae -Associated Necrotizing Pneumonia in Children: a case-report - Chan ho Lee, So Yoon Jo, Keon Woo Na, Sung Won Kim, Yoon Ha Hwang
-
Kosin Medical Journal 2019;34(1):57-64.
DOI: https://doi.org/10.7180/kmj.2019.34.1.57
Published online: June 30, 2019
Department of Pediatrics, Busan St. Mary's Hospital, Busan, Korea.
- Corresponding Author: Yoon Ha Hwang, Department of Pediatrics, Busan St. Mary's Hospital, 25–14, Yongho-ro, 232beon-gil, Nam-gu, Busan 48575, Korea. Tel: +82-51-933-7986, Fax: +82-51-932-8600, hyh190@naver.com
• Received: June 14, 2018 • Revised: October 1, 2018 • Accepted: October 1, 2018
Copyright © 2019 Kosin University College of Medicine
Articles published in Kosin Medical Journal are open-access, distributed under the terms of the Creative Commons Attribution Non-Commercial License (http://creativecommons.org/licenses/by-nc/4.0/) which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
- 1,665 Views
- 7 Download
- 1 Crossref
Abstract
- Mycoplasma pneumoniae is the most common bacterial strain causing atypical pneumonia in children and adolencents. Although it is known to cause mild symptoms, it can also cause severe pulmonary or extrapulmonary complications in rare cases. Necrotizing pneumonia (NP) is often reported as a complication of Streptococcus pneumoniae and is very rarely caused by M. pneumoniae. We report a case in which a 5-year-old boy was diagnosed with lobar pneumonia with symptoms that aggravated even with macrolide antibiotic treatment. Anti-mycoplasma pneumoniae Ig-M test yielded high values, and direct polymerase chain reaction results were also positive. NP caused by M. pneumoniae was confirmed on computed tomography. After treatment involving tosufloxacin and systemic steroid, the lesion decreased in size and improved gradually when followed-up for more than 1 year. The patient did not have any predisposing or risk factors for NP.
- A 5-year-old boy who was otherwise healthy and had no underlying disease was diagnosed with pneumonia and hospitalized for 10 days for treatment at a local clinic. Although the patient was treated with clarithromycin, one of a macrolide antibiotic, for 10 days, his fever and dry coughs worsened, and aggravation of pneumonia was also confirmed on chest x-ray. The patient did not exhibit hemoptysis. He did not have any gastrointestinal symptom, chest pain, rash, or arthralgia. The patient had not travelled recently. His past medical history, social history, or family history did not show any particular things to remark. According to physical examination conducted at admission, the body temperature was 37.8℃, the heart rate was 128/min, and the breathing rate was 22/min. Oxygen saturation was 97% in atmosphere. The patient showed acute ill-looking appearance. Although lower left lung sounds were diminished compared with the right side, rale was not noted. The patient had normal heart sounds, and no heart murmur was detected.
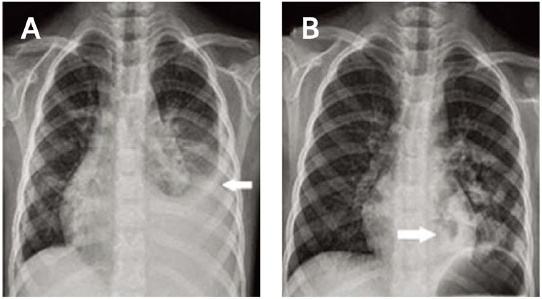
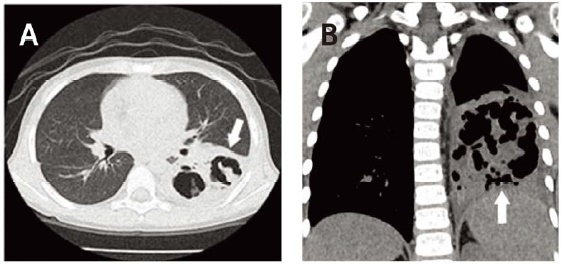
- According to the blood test conducted at admission, the patient had a white blood cell count of 20,010/mm3 (neutrophil 85%, lymphocyte 8%), a hemoglobin count of 10.8 g/dL, and a platelet count of 242,000/µL. In addition, aspartate aminotransferase (AST), alanine aminotransferase (ALT), lactate dehydrogenase (LDH) were mildly elevated. C-reactive protein (CRP) and erythrocyte sedimentation rate (ESR) were also elevated to 169.18 mg/dL, and 19 mm/h, respectively (Table 1). Nested polymerase chain reaction (PCR) conducted on the day of admission to identify the bacteria causing pneumonia showed positive result for M. pneumoniae but negative for Streptococcus pneumoniae, Haemophilus influenzae, Legionella pneumophila, Chlamydophila pneumoniae, and Bordetella pertussis. IgM and IgG titers against M. pneumoniae were positive at 3,719 U/mL (0–770 U/mL) and 1.05 (index < 0.9), respectively. The results of real-time PCR conducted to identify on respiratory viruses were negative for adenovirus, respiratory syncytial virus, influenza, parainfluenza, bocavirus, metapneumovirus, and rhinovirus, but positive for coronavirus. The patient was regarded as co-infected with respiratory virus. It was considered that the co-infection of the virus in the case of M. pneumoniae pneumonia may exacerbate the clinical course of the pneumonia and the severity of the disease.5 The results of blood and urine cultures were all negative. On chest x-ray conducted the day of admission, pneumonic infiltration of both lung fields and pleural effusion of the left lung field were noted (Fig. 1A). Because he was suspected to have bacterial pneumonia with severe inflammation, based on the result of his blood tests & PCR, cefuroxime was administered intravenously and clarithromycin also continued. His symptoms and chest x-ray results were progressing in spite of the use of clarithromycin, one of the widely used macrolide antibiotic for M. pneumoniae, for 15 days on Day 5 of admission. The macrolide antibiotic was changed to azthromycin and 2 mg/kg/day (divided into 2 doses/day) of methylprednisolone as a systemic steroid were administered intravenously.267 Fever subsided on the second day after antibiotic replacement and steroid administration. Then the dry cough became productive of sputum. The overall systemic condition also improved. A blood test conducted on Day 6 of admission revealed that AST and ALT had decreased to 26 U/L and 146 U/L, respectively, whereas LDH was elevated to 864 U/L. Moreover, CRP and ESR had decreased to 4 mg/dL and 21 m/h, respectively (Table 1). On Day 8 of admission, High fever recurred and cough productive of sputum increased in frequency. Severe respiratory symptoms continued until Day 10 of admission. We suspected macrolide-resistant M. pneumoniae pneumonia because fever continued without improvement of symptoms even with treatment with macrolide antibiotics. In addition, complications of pneumonia, such as pleural effusion, were also suspected because the fever continued after appropriate treatment with macrolide antibiotics. Chest x-ray was performed, revealing pneumonic infiltration of the left lung accompanied by multiple cavities in the lower lobe of the left lung (Fig. 1B). To accurately evaluate NP, we performed chest computed tomography (CT), confirming lobar fibrosis with multiple air-filled cavities in the left lower lobe as well as pleural effusion at the site of pneumonic infiltration (Fig. 2A, 2B). Therefore, the antibiotic was changed to 10 mg/kg/day (divided into 2 doses/day) of tosufloxacin, a quinolone antibiotic.6 After 15 days of administration of methylprednisolone, the dose was gradually tapered for 7 days. Tosufloxacin was administered for 10 days. Severe M. pneumoniae pneumonia may occasionally be associated with changes in lung function and chronic lung complications. A pulmonary function test conducted on Day 50 after the onset revealed that FVC, FEV1 and FEF25–75 were diminished to 0.94 L (61%), 0.84 L (61%), and 1.12 L/s (63%), respectively, which was consistent with reduced pulmonary function. Thereafter, no additional repeat pulmonary function tests were performed. After 8 months of follow-up, the patient improved gradually without surgery or medication.
CASE
- M. pneumoniae is a major cause of CAP in children, and strains that are resistant to macrolide are now commonly observed. M. pneumoniae is often observed in young adults and those above the age of 5 years, and particularly in immunocompromised individuals such as those with HIV, those who have used steroids for long periods of time, those having anti-cancer therapy, those with sickle cell diseases, smokers, and those with chronic lung diseases.8
- Delayed administration of appropriate antibiotics, regardless of antibiotic resistance, may cause symptoms to persist.9 A number of severe pulmonary complications have been report with M. pneumoniae infection. NP is a complication of lobar pneumonia characterized by necrosis and liquefaction of the lung tissue.10 NP can present with various symptoms, such as fever, cough, chest pain, and dyspnea, and is diagnosed through chest CT.11 If protracted course of fever or worsening respiratory exertion occur in patients with M. pneumoniae pneumonia, the possibility of complicated parapneumonic effusion or NP should be considered. The pathogenesis of M. pneumoniae-associated necrotizing pneumonia is still not clear. However, it onset might be associated with the state of immunity of the hosts because the severity of M. pneumoniae pneumonia is also closely related to the patient immune status.11 Macrolide-resistent M. pneumoniae infection, which is increasingly being reported, also might be another reason for the severe M. pneumoniae pneumonia. Secondary bacterial infection after M. pneumoniae pneumonia may also contribute to severe infection.11
- Conservative management of NP with antibiotics and chest drainage for pleural effusions results in good outcomes, and the surgical resection is rarely performed. Though pleural effusion associated with M. pneumoniae infection was usually considered as small amount and being self-limited, large pleural effusions require chest tube insertion and drainage. Because lung development continues after birth until 8 years of age, most pediatric patients with NP recover without surgical intervention regardless of the pathogen. Early recognition and treatment with appropriate antibiotics may lead to complete resolution of the cavity lesion. But lung resection can be considered as an alternative treatment option in patients who are unresponsive to antibiotic therapy and develop parenchymal complications.212
- In the present case, Macrolide-resistent M. pneumoniae pneumonia, without any specific risk factor, that did not respond to appropriate antibiotic treatment progressed to NP. We performed a chest CT when the cavities at the site of the pneumonia were seen on chest x-ray on Day 20 after onset. Minimal parapneumonic effusion and cavities was detected; therefore thracocentesis and chest tube insertion were not performed.
- Macrolide antibiotics is the first choice agent against M. pneumoniae pneumonia, and doxycycline and fluoroquinolone can also be used as second choice.3,6,10 Because quinolone can cause resistance in bacteria other than M. pneumoniae and can cause musculoskeletal side effects, such as rupture of tendons, arthralgia, and tendinitis, it should be used with caution in children and adolescents.513 If fever continues without improvement of the symptoms even with treatment with macrolide antibiotics, macrolide-resistent M. pneumoniae pneumonia should be suspected.9 Quinolone antibiotics can be used to treat Macrolide-resistent M. pneumoniae pneumonia.2914
- Steroids can also be used if macrolide-resistent M. pneumoniae pneumonia progresses to severe pneumonia8914 Hyperimmune reactions of T-cell which was already sensitized by previous M. pneumoniae infection are thought to flood the lung tissue, and produce a variety of inflammatory mediatory mediators.215 From the rapid improvements of clinical symptoms and pulmonary lesions in the severe M. pneumoniae pneumonia patients treated corticosteroids, it has been proposed that cell-mediated immunity plays an important role in the progress of M. pneumoniae pneumonia. Immunosuppressive therapy, such as corticosteroids, has ability to downregulate the cell-mediated immune response, and has shown a profound effect by reducing the immune-mediated pulmonary injury seen in severe M. pneumoniae infection. Clinical experiences and animal model suggested that the use of both antimicrobial therapy and corticosteroids is important to improve the outcome of severe M. pneumoniae pneumonia.28911 Therefore, we had used systemic steroids for a short period of time.
- Serum interleukin (IL)-18 is believed to be associated with the severity of pneumonia because its level increases particularly in the acute phase and it induces the expression of various cytokines.15 Thus, The serum IL-18 level was thought to be a useful as a predictor of refractory or severe M. pneumoniae pneumonia. The serum levels of IL-18 correlated significantly with those of lactate dehydrogenase (LDH).1115 In this case, patient was treated with clarithromycin, a widely used macrolide antibiotic, for 10 days, but the fever and respiratory symptoms persisted after the treatment. The serum LDH level was elevated by more than 400 U/L. Therefore, we suspected macrolide-resistent M. pneumoniae pneumonia and changed the antibiotics to tosufloxacin, which was continued for 10 days. We also administered systemic steroid treatment for 17 days, and tapered after which the symptoms improved.
- Our case has two limitations. First, Microbiological laboratory tests was not performed. For detection of macrolide-resistent M. pneumoniae, a search for mutation at site 2063, 2064 and 2617 in the M. pneumonia 23S rRNA domain V gene region shoud be performed using a direct sequencing method in samples with a positive PCR result and the minimum inhibitory concentration of antimicrobial agents for the M. pneumoniae isolates should be determined by micro-dilution methods. Recently, the 23S rRNA mutations of mycoplasma using PCR have been reported, it has not yet been established as a clinical trial. We need to research on the guidelines for application to clinical practice. Second, follow up evaluation after discharge was not performed to assess the recovery of NP without surgical intervention after discharge.
- M. pneumoniae-associated pneumonia sometimes progresses to severe pneumonia. To prevent complications, early investigation into antibiotic resistance is helpful to allow for an appropriate use of antibiotics as well as combined use of systemic steroids. Therefore, additional studies on diagnostic tests for macrolide-resistent M. pneumoniae pneumonia are required, and studies should also investigate indications and side effects of quinolone antibiotics to ensure their appropriate use.
DISCUSSION
- 1. Ferwerda A, Moll HA, de Groot R. Respiratory tract infections by Mycoplasma pneumoniae in children: a review of diagnostic and therapeutic measures. Eur J Pediatr 2001;160:483–491.ArticlePubMed
- 2. Lee NY, Hur TH, Song SW, Lee HK, Lee KY, Lee HS, et al. Clinical Aspects of Necrotizing Pneumonitis Resulting from Mycoplasma Pneumoniae Infection in Children. Pediatr Allergy Respir Dis 2007;17:183–195.
- 3. Hacimustafaoglu M, Celebi S, Sarimehmet H, Gurpinar A, Ercan I. Necrotizing pneumonia in children. Acta Paediatr 2004;93:1172–1177.ArticlePubMed
- 4. Kawai Y, Miyashita N, Kubo M, Akaike H, Kato A, Nishizawa Y, et al. Nationwide surveillance of macrolide-resistant Mycoplasma pneumoniae infection in pediatric patients. Antimicrob Agents Chemother 2013;57:4046–4049.ArticlePubMedPMC
- 5. Kim JH, Kim EJ, Kwon JH, Seo WH, Yoo Y, Choung JT, Song DJ. Clinical characteristics of respiratory viral coinfection in pediatric Mycoplasma pneumoniae pneumonia. Allergy Asthma Respir Dis 2017;5:15–20.Article
- 6. Committee for the Guidelines in Management of Respiratory Infectious Diseases in Children. In: Ouchi K, Kurosaki T, Okada K, editors. Guidelines for the management of respiratory infectious diseases in children in Japan. Japanese Society of Pediatric Pulmonology; Japanese Society for Pediatric Infectious Diseases; 2011.
- 7. Tamura A, Matsubara K, Tanaka T, Nigami H, Yura K, Fukaya T. Methylprednisolone pulse therapy for refractory Mycoplasma pneumoniae pneumonia in children. J Infect 2008;57:223–228.ArticlePubMedPMC
- 8. Morozumi M, Takahashi T, Ubukata K. Macrolideresistant Mycoplasma pneumoniae: characteristics of isolates and clinical aspects of community-acquired pneumonia. J Infect Chemother 2010;16:78–86.ArticlePubMed
- 9. Klement E, Talkington DF, Wasserzug O, Kayout R, Davidovitch N, Dumke R, et al. Identification of risk factors for infection in an outbreak of Mycoplasma pneumoniae respiratory tract disease. Clin Infect Dis 2006;43:1239–1245.ArticlePubMed
- 10. Chiu CY, Chiang LM, Chen TP. Mycoplasma pneumonia infections complicated by necrotizing pneumonitis with massive pleural effusion. Eur J Pediatr 2006;165:275–277.ArticlePubMed
- 11. Ma YJ, Wang SM, Cho YH, Shen CF, Liu CC, Chi H, et al. Clinical and epidemiological characteristics in children with community-acquired mycoplasma pneumonia in Taiwan: A nationwide surveillance. J Microbiol Immunol Infect 2015;48:632–638.ArticlePubMed
- 12. Sawicki GS, Lu FL, Valim C, Cleveland RH, Colin AA. Necrotising pneumonia is an increasingly detected complication of pneumonia in children. The European respiratory journal 2008;31:1285–1291.ArticlePubMed
- 13. Maroushek SR. Principles of antimycobacterial therapy. In: Kliegman RM, Stanton BF, St. Geme JW, Schor NF, Behrman RE, editors. Nelson textbook of pediatrics. 20th ed. Philadelphia: Elsevier; 2105. p. 1439–1444.
- 14. Wy HH, Min DH, Kim DS, Park MS, Shim JW, Jung HL, et al. Clinical characteristics of Mycoplasma pneumoniae pneumonia in Korean children during the recent 3 epidemics. Allergy Asthma Respir Dis 2017;5:8–14.Article
- 15. Miyashita N, Kawai Y, Inamura N, Tanaka T, Akaike H, Teranishi H, et al. Setting a standard for the initiation of steroid therapy in refractory or severe Mycoplasma pneumoniae pneumonia in adolescents and adults. J Infect Chemother 2015;21:153–160.ArticlePubMed
References
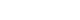
Fig. 1
(A) Performed on the day of admission, showing pneumonic infiltration of both lung fields as well as pleural effusion of the left lung field (white arrow).
(B) Performed on Day 10 of admission, showing slight improvement in pneumonic consolidation in both lung fields, and cavity formation (white arrow) in both the left lower lung and retrocardiac space.
Chest x-ray findings of case

Figure & Data
References
Citations
Citations to this article as recorded by 

- Cavitatory necrotising pneumonia with extrapulmonary multi-systemic involvement — a rare presentation of macrolide-resistant Mycoplasma pneumoniae
Namitha Mohan, Vishal Dnyaneshwar Sawant, Alpana Kondekar
The Egyptian Journal of Internal Medicine.2023;[Epub] CrossRef

 KOSIN UNIVERSITY COLLEGE OF MEDICINE
KOSIN UNIVERSITY COLLEGE OF MEDICINE
 PubReader
PubReader ePub Link
ePub Link Cite
Cite